This week I’ve been on the Gold Coast for the 12th Mosquito Control Association of Australia and Arbovirus Research in Australia Symposium. The theme of the meeting was “Managing challenges and threats with new technology” and included presentations covering a range of topics, from remote piloted aircraft for mosquito control to the discovery of insect-specific viruses and their potential to stop outbreaks of mosquito-borne disease.
You can check out some of the tweets shared during the meeting here.
I found myself on ten papers presented at the meeting and I’ve provided the abstracts below!
Does surrounding land use influence the mosquito populations of urban mangroves?
Suzi B. Claflin1 and Cameron E. Webb2,3
1Department of Entomology, Cornell University, Ithaca, NY, USA; 2Marie Bashir Institute of Infectious Diseases and Biosecurity, University of Sydney, Sydney, NSW; 3Department of Medical Entomology, NSW Health Pathology, Westmead Hospital, NSW 2145, Australia
Mosquitoes associated with mangrove habitats pose a pest and public health risk. These habitats in urban environments are also threatened by urbanisation and climate change. As a consequence, urban mangrove management must strike a balance between environmental conservation and minimising public health risks. Land use may play a key role in shaping the mosquito community within urban mangroves through either species spillover or altering the abundance of mosquitoes associated with the mangrove. In this study, we explore the impact of land use within 500m of urban mangroves on the abundance and diversity of adult mosquito populations. Carbon dioxide baited traps were used to sample host-seeking female mosquitoes around nine mangrove forest sites along the Parramatta River, Sydney, Australia. Specimens were identified to species and for each site, mosquito species abundance, species richness and diversity were calculated and were analyzed in linear mixed effects models. We found that the percentage of residential land and bushland in the surrounding area had a negative effect on mosquito abundance and species richness. Conversely, the amount of mangrove had a significant positive effect on mosquito abundance, and the amount of industrial land had a significant positive effect on species richness. These results demonstrate the need for site-specific investigations of mosquito communities to assist local authorities develop policies for urban development and wetland rehabilitation.
Do urban wetlands increase mosquito-related public health risks?
Jayne K. Hanford1, Cameron E. Webb2,3, Dieter F. Hochuli1
1 School of Life and Environmental Sciences, The University of Sydney, Sydney; 2 Medical Entomology, NSW Health Pathology, Level 3 ICPMR, Westmead Hospital, Westmead; 3Marie Bashir Institute of Infectious Diseases and Biosecurity, The University of Sydney, Sydney
Wetlands in urban areas are frequently constructed or rehabilitated to improve stormwater quality and downstream aquatic health. In addition to improving water quality, these wetlands can provide aesthetic, recreational and biodiversity values to communities. However, urban wetlands are often perceived to proliferate nuisance-biting and pathogen-transmitting mosquitoes which can, in severe cases, erode goodwill in the community for protecting these valuable ecosystems. We compared mosquito assemblages at 24 natural and constructed wetlands in the greater Sydney region, Australia. Our aims were to determine if wetlands with high aquatic biodiversity posed reduced mosquito-related public health risks, and if these links vary across the urban-rural gradient. At each wetland we sampled adult and larval mosquitoes, aquatic macroinvertebrates and physical habitat variables on two occasions through summer and autumn. Although larval mosquito abundance was low across all sites, there was a high diversity of adult mosquito species, and assemblages varied greatly between sites and seasons. Species of wetland-inhabiting mosquitoes showed vastly different responses to aquatic biodiversity and physical habitat variables. There were strong relationships between the abundance of some mosquito species and aquatic macroinvertebrate richness, while others mosquito species showed strong relationships with the percentage of urbanisation surrounding the wetland. Effectively integrating wetlands into cities requires balancing wetland design for water infrastructure purposes, biodiversity resources and public health and wellbeing requirements. Understanding relationships between biodiversity value and mosquito-related public health risks will enhance the value of constructed urban wetlands in cities while minimising risks posed by mosquitoes.
Aedes aegypti at Sydney Airport; the detections and response
Doggett, S.L. and Webb C.E
Department of Medical Entomology, CIDMLS, Pathology West, ICPMR,
Westmead Hospital, Westmead, NSW.
Despite a huge increase in the detections of exotic vectors at ports around Australia, up until 2016 there had been no detection of Aedes aegypti at the Sydney International Airport. However, this changed on 14/Jan/2016 when two larvae were observed in an ovitrap serviced by the Department of Agriculture and Water Resources (formerly AQIS), as part of their routine surveillance activities for the detection of exotic vectors. These larvae were confirmed as being Ae. aegypti. Thereafter, there were a further nine separate detections of Ae. aegypti up until 4/Mar/2016. Six were via BG traps, one in an ovitrap, and there were two separate instances of an adult mosquito being collected in open areas. The majority of detections occurred in areas of the airport known as the ‘basement areas’. This is where the bags are unloaded from the air cans onto convey belts for collection directly upstairs by the passengers. Response measures undertaken included: (1) enhanced surveillance; BG traps were increased in number from 2 to 12, and traps inspected at more frequent intervals; (2) insecticide treatments; thermal fogging and surface sprays were conducted of the relevant areas; (3) vector surveys; a comprehensive audit of the airport was undertaken to examine the potential for localized mosquito breeding. In the case of the vector surveys, some 107 potential sites were identified and grouped into risk categories. No Ae. aegypti were discovered breeding, although Cx. quinquefasciatus and Ae. notoscriptus were found, and recommendations to prevent future localized breeding were made.
Communicating the risks of local and exotic mosquito-borne disease threats to the community through social and traditional media
Cameron E Webb1,2
1Department of Medical Entomology, NSW Health Pathology, Level 3, ICPMR, Westmead Hospital, WESTMEAD NSW 2145 AUSTRALIA; 2Marie Bashir Institute of Infectious Diseases and Biosecurity, University of Sydney, NSW 2006, AUSTRALIA
Mosquito-borne disease management in Australia faces challenges on many fronts. Many gaps exist in our understanding of the drivers of exotic and endemic mosquito-borne disease risk but also the pathways to ensuring the community embrace personal protection measures to avoid mosquito bites. While traditional media has been the mainstay of public health communications by local authorities, social media provides a new avenues for disseminating information and engaging with the wider community. This presentation will share some insights into how the use of social media has connected new and old communications strategies to not only extend the reach of public health messages but also provide an opportunity to promote entomological research and wetland conservation. A range of social media platforms, including Twitter, Instagram and WordPress, were employed to disseminate public health messages and engage the community and traditional media outlets. Engagement with the accounts of traditional media (e.g. radio, print, television, online) was found to be the main route to increased exposure and, subsequently, to increased access of public health information online. With the increasing accessibility of the community to online resources via smartphones, researchers and public health advocates must develop strategies to effectively use social media. Many people now turn to social media as a source of news and information and those in the field of public health, as well as entomological research more generally, must take advantage of these new opportunities.
See the slides here.
So, you want to write a field guide?
Cameron E. Webb1,2, Stephen L. Doggett1 and Richard C. Russell2
1Department of Medical Entomology, NSW Health Pathology, Level 3, ICPMR, Westmead Hospital, WESTMEAD NSW 2145 AUSTRALIA; 2Marie Bashir Institute of Infectious Diseases and Biosecurity, University of Sydney, NSW 2006, AUSTRALIA
We know a lot about Australian mosquitoes. They’re one of the most studied insects in the country. Their pest and public health threats warrant a better understanding of their biology and ecology. There is still plenty we don’t know. We may not understand their ecological role in the local environment very well and there are many mosquitoes we know exist but have very little information about them. We still need to give many mosquitoes a formal scientific name. There is a reason why so many field guides are written by retired scientists. It’s not just about expertise, it’s about time too! In early 2016, “A Guide to Mosquitoes of Australia” to was published by CSIRO Publishing and marked the culmination of many years work. This work involved chasing mosquitoes from coastal rock pools to snow melt streams. We carried eskies of buzzing mosquitoes on airplanes from northern Australia to laboratories in Western Sydney and there were many late nights of wrangling those mosquitoes to get the perfect photo. Lots of mosquito bites too. Many, many mosquito bites. Putting together this field guide wasn’t an easy task and for all those involved it proved a challenge in many different ways. Digging out old papers to colour-correcting digital photographs proved time consuming but the biggest delays in finishing this project was a problem that plagues many field guide writer, “species creep”! Completing the guide was only possible with the kindness, generosity and co-operation of many mosquito researchers around the country.
See the slides here.
Arbovirus and vector surveillance in NSW, 2014/15-2015/16
Doggett, S.L., Clancy, J., Haniotis, J., Webb C. and Toi, C.
Department of Medical Entomology, CIDMLS, Pathology West, ICPMR,
Westmead Hospital, Westmead, NSW.
The NSW Arbovirus Surveillance and Vector Monitoring Program acts as an early warning system for arbovirus activity. This is achieved through the monitoring of mosquito abundance, detection of arboviruses from mosquitoes, and the testing for seroconversions to MVEV and KUNV in sentinel chickens. A summary of the last two seasons will be presented. The 2014-2015 season started early with elevated temperatures through late 2014, however conditions were relative dry with neither Forbes’ nor the Nicholls’ hypothesis being suggestive of an MVEV epidemic. Despite this, for the inland region, human notifications were close to average, with 260RRV & 11BFV). There were 12 arboviral detections from the inland including 5BFV, 6RRV & 1STR, with no seroconversions. In contrast, the coastal strip experienced the largest epidemic of RRV in recorded history. The 1,225 cases were close to double the average, with much of the activity occurring in the far north coast. There were 41 isolates from the mosquitoes trapped along the coast and included 6BFV, 29RRV, 4EHV and 2STRV. An intense El Niño occurred during the 2015-2016 season and thus it was extremely dry across the state. Again the Forbes’ and the Nicholls’ hypothesis were not suggestive of an MVEV outbreak. For the inland, mosquito numbers were well below average and there were only two arboviral detections from the mosquitoes (1RRV & 1 BFV), with no seroconversions. Similarly, mosquito collections were below average and there were also two arboviral detections from the trapped mosquitoes (1BFV & 1EHV). Human cases were below average.
Are remote piloted aircraft the future of mosquito control in urban wetlands?
Cameron E Webb1,2 Stephen L. Doggett1 and Swapan Paul3
1Department of Medical Entomology, NSW Health Pathology, Level 3, ICPMR, Westmead Hospital, WESTMEAD NSW 2145 AUSTRALIA; 2Marie Bashir Institute of Infectious Diseases and Biosecurity, University of Sydney, NSW 2006, AUSTRALIA; 3Sydney Olympic Park Authority, 8 Australia Ave, Sydney Olympic Park NSW 2127, AUSTRALIA
Mosquito control in urban wetlands will become increasing important. The expansion of residential areas will continue to encroach on natural mosquito habitats, particularly coastal wetlands, and expose the community to the health risks associated with mosquitoes. In many existing areas, ever increasing density of human populations associated with high rise residential developments will further expose people to mosquitoes. The increasing urban development adjacent to wetlands can restrict the ability to use traditional larvicide and insect growth regulator application methods. In 2016 a trial of larvicide application via remote piloted aircraft was undertaken in an area of estuarine wetlands at Sydney Olympic Park. An existing mosquito control program involving helicopter application of larvicides has been in place for over a decade. Post-treatment mortality of Aedes vigilax and Culex sitiens larvae was compared between bioassay and long-term surveillance sites within the wetlands. While there was a substantial reduction in larval densities post-treatment, the treatments via remote piloted aircraft were less effective than those of traditional piloted aircraft. The results of this preliminary trial suggest that the use of remote piloted aircraft has potential but the operational aspects of this application method requires careful consideration if there are to be as effective as existing strategies.
Seasonal Activity, Vector Relationships and Genetic Analysis of Mosquito-Borne Stratford Virus
Cheryl S. Toi1, Cameron E. Webb1,2, John Haniotis1, John Clancy1 and Stephen Doggett1
1Department of Medical Entomology, Centre for Infectious Diseases and Microbiology Laboratory Services, Pathology West – Institute for Clinical Pathology and Medical Research, Westmead Hospital, NSW; 2Marie Bashir Institute for Infectious Diseases and Biosecurity, University of Sydney, Institute for Clinical Pathology and Medical Research, Westmead Hospital, NSW;
There are many gaps to be filled in our understanding of mosquito-borne viruses, their relationships with vectors and reservoir hosts, and the environmental drivers of seasonal activity. Stratford virus (STRV) belongs to the genus Flavivirus and has been isolated from mosquitoes and infected humans in Australia. However, little is known of its vector and reservoir host associations. A total of 43 isolates of STRV from field collected mosquitoes collected in NSW between 1995 and 2013 were examined to determine the genetic diversity between virus isolates and their relationship with mosquito species by year of collection. The virus was isolated from six mosquito species; Aedes aculeatus, Aedes alternans, Aedes notoscriptus, Aedes procax Aedes vigilax, and Anopheles annulipes. While there were distinct differences in temporal and spatial activity of STRV, with peaks of activity in 2006, 2008, 2010 and 2013, there was a high degree of sequence homology (89.1% – 97.7%) found between isolates with no evidence of mosquito species, geographic, or temporal divergence. The result suggests the virus is geographically widespread in NSW (albeit only from coastal regions) and increased local STRV activity is likely to be driven by reservoir host factors and local environmental conditions influencing vector abundance. While STRV may not currently be associated with major outbreaks of human disease, with the potential for urbanisation and climate change to increase mosquito-borne disease risks, and the potential for genomic changes which could produce pathogenic strains, understanding the drivers of STRV activity may assist the development of strategic response to public health risks posed by zoonotic flaviviruses in Australia.
Insect specific flaviviruses suppress West Nile virus replication and transmission
Sonja Hall-Mendelin1, Breeanna McLean2, Helle Bielefeldt-Ohmann3, Cameron E. Webb4 Jody Hobson-Peters2, Roy Hall2, Andrew van den Hurk1
1Public Health Virology, Forensic and Scientific Services, Department of Health, Queensland Government, PO Box 594, Archerfield 4108, Queensland, Australia; 2Australian Infectious Diseases Research Centre, School of Chemistry and Molecular Biosciences, The University of Queensland, St Lucia 4072, Queensland, Australia; 3School of Veterinary Science, The University of Queensland, Gatton Campus, Gatton 4343, Queensland, Australia; 4Medical Entomology, Marie Bashir Institute of Infectious Diseases and Biosecurity, The University of Sydney, NSW, Australia
Diseases caused by mosquito-borne flaviviruses, including dengue (DENV), Zika and West Nile viruses (WNV), are a global problem. New molecular tools have led to recent discoveries of a plethora of insect-specific flaviviruses (ISF) that infect mosquitoes but not vertebrates. Preliminary reports have suggested that transmission of WNV can be suppressed by some ISFs in co-infected mosquitoes, thus the ecology of ISFs and their potential as natural regulators of flaviviral disease transmission is intriguing. In vitro studies with two ISFs discovered in Australia, Palm Creek virus (PCV) and Parramatta River virus (PaRV), demonstrated suppression of WNV, Murray Valley encephalitis virus (MVEV) and DENV replication in mosquito cells (C6/36) previously infected with either of these ISFs. Further in vivo experiments indicated that these ISFs were not transmitted horizontally in the saliva, and that PaRV relied on vertical transmission through the mosquito egg to the progeny. Additional studies revealed a significant reduction of infection and transmission rates of WNV when Culex annulirostris were previously infected with PCV, compared to control groups without PCV. Of particular interest was the specific localisation of ISFs to the midgut epithelium of mosquitoes infected via natural route (vertical transmission – PaRV) or by intrathoracic injection (PCV). Overall these results confirm a role for ISFs in regulating the transmission of pathogenic flaviviruses by mosquitoes and that this interference may occur in the midgut where initial infection occurs. Further research is needed to determine the precise mechanism of this phenomenon and its potential for mosquito-borne disease management.
Neges, Nidos and Nings – so that’s what’s killing my mossie cells!
Roy Hall1, Jody Hobson-Peters1, Helle Bielefeldt-Ohmann1, Caitlin O’Brien1, Breeanna McLean1, Agathe Colmant1, Jessica Harrison1, Thisun Piyasena1, Natalee Newton1, Waylon Wiseman1, Marcus Mah1,2, Natalie Prow2, Andreas Suhrbier2, David Warrilow3, Andrew van den Hurk3, Sonja Hall-Mendelin3, Cheryl Johansen4, Steven Davis5, Weng Chow6, Stephen Doggett7, John Haniotis7 and Cameron Webb7.
1Australian Infectious Diseases Research Centre, School of Chemistry and Molecular Biosciences, The University of Queensland, Australia; 2QIMR Berghofer Medical Research Institute, Herston, Brisbane, Australia; 3Public Health Virology, Forensic and Scientific Services, Coopers Plains, Queensland, Australia; 4Arbovirus Surveillance and Research, Infectious Diseases Surveillance Unit, PathWest Laboratory Medicine WA, Western Australia; 5Berrimah Veterinary Laboratories, Department of Primary Industry and Fisheries, Darwin, Northern Territory, Australia; 6Vector Surveillance and Control, Australian Army Malaria Institute, Enoggera, Queensland, Australia; 7Department of Medical Entomology, West Westmead Hospital, Westmead, NSW, Australia.
Isolation of viruses from mosquitoes is an important component of arbovirus surveillance and virus discovery programs. In our lab, these viruses are detected in inoculated cultures by the appearance of cytopathic effects (CPE) in mosquito cell monolayers or by reactivity of monoclonal antibodies to viral antigens or dsRNA intermediates. Isolates are then identified by RT-PCR or deep sequencing. We detected extensive CPE in many mosquito cell cultures inoculated with mosquito homogenates from several regions of Australia, however these isolates were not identified by specific mAbs or RT-PCRs designed to detect known arboviruses. When we investigated their identity by deep sequencing, a new species (Castlerea virus – CsV) in the unclassified taxon Negevirus, was identified in several mosquito species from WA and Brisbane. Two viruses in the newly established Mesoniviridae family (order Nidovirales) were also identified; a novel species named Casuarina virus (CASV) from Coquillettidia xanthogaster in Darwin and from Culex annulirostris in Cairns, and the first Australian isolates of Nam Dinh virus from several mosquito species in Brisbane and Perth. Many isolates of a new genetic lineage of Liao Ning virus, a member of the Seadornavirus genus (family Reoviridae), were also obtained from several mosquito species from different regions of Australia. These new viruses were isolated at very high frequency in some mosquito collections, and were often found to co-infect isolates of other mosquito-borne viruses making it difficult to obtain pure cultures. We have now developed neutralising antibodies to each virus to facilitate selective removal of these viruses from mixed cultures.
That was a busy meeting. I’m exhausted but cannot wait until the next meeting in 2018. Are you a member of the Mosquito Control Association of Australia?





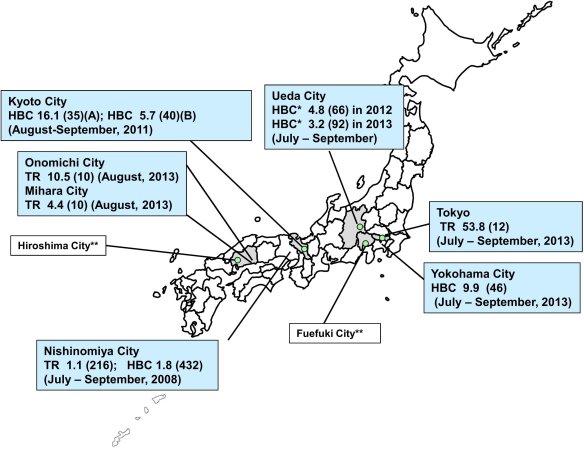
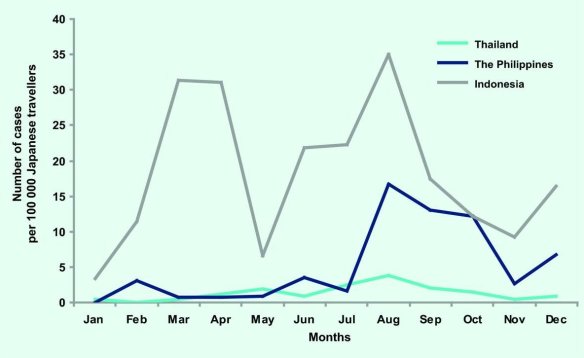
 Considered free of dengue for around 70 years, Japan is now facing an outbreak of mosquito-borne dengue virus centered around a popular Tokyo city park. How could this happen?
Considered free of dengue for around 70 years, Japan is now facing an outbreak of mosquito-borne dengue virus centered around a popular Tokyo city park. How could this happen?